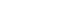UCLA engineers develop branch-and-leaves design for super-efficient electrodes
Staff
Electronics supercapacitor UCLAThe more ions an electrode exchanges, the faster it delivers power. UCLA engineers designed their electrode with this in mind, maximizing its surface area, creating the most possible space for it to attract electrons.
With automakers focusing more and more on developing hybrid vehicles and regenerative braking systems, the need to develop a highly efficient rechargeable energy storage device is on the minds of researchers and mechanical engineers.

The branch-and-leaves design is made up of arrays of hollow, cylindrical carbon nanotubes (the ‘branches’) and sharp-edged petal-like structures (the ‘leaves’) made of graphene. Photo courtesy of UCLA Engineering.
A team from UCLA Henry Samueli School of Engineering and Applied Science and four other institutions have designed a super-efficient and long-lasting electrode for supercapacitors.
Mechanical engineers designed the device based on the structure and function of leaves on tree branches, which are able to absorb ample amounts of carbon dioxide for photosynthesis because of the surface area of their leaves.
“We often find inspiration in nature, and plants have discovered the best way to absorb chemicals such as carbon dioxide from their environment,” said Tim Fisher, the study’s principal investigator and a UCLA professor of mechanical and aerospace engineering. “In this case, we used that idea but at a much, much smaller scale — about one-millionth the size, in fact.”
The electrode design provides the same amount of energy storage and delivers as much power as other standard electrodes. However, the particular design is much smaller, lighter and it is more than 10 times more efficient than other designs.
In experiments it produced 30 percent better capacitance for its mass compared to the best available electrode made from similar carbon materials, and 30 times better capacitance per area. It also retained 95 percent of its initial capacitance after more than 10,000 charging cycles.
One of the challenges the engineers faced with this supercapacitor project was producing more efficient and durable electrodes. Electrodes attract ions, which store energy, to the surface of the supercapacitor, where that energy becomes available to use. Ions in supercapacitors are stored in an electrolyte solution. An electrode’s ability to deliver stored power quickly is determined in large part by how many ions it can exchange with that solution: The more ions it can exchange, the faster it can deliver power.
It is with this understanding that the researchers designed their electrode — maximizing its surface area — creating the most possible space for it to attract electrons.
In order to develop the branch-and-leaves design, the researchers used two nanoscale structures composed of carbon atoms. The “branches” are arrays of hollow, cylindrical carbon nanotubes, about 20 to 30 nanometers in diameter; and the “leaves” are sharp-edged petal-like structures, about 100 nanometers wide, that are made of graphene. The leaves are then arranged on the perimeter of the nanotube stems. The leaf-like graphene petals also give the electrode stability.
The engineers then formed the structures into tunnel-shaped arrays, which the ions that transport the stored energy flow through with much less resistance between the electrolyte and the surface to deliver energy than they would if the electrode surfaces were flat.
Their work is described in the journal Nature Communications.