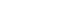Researchers create ultra-thin “Diamond” nano-thread
By Design Engineering Staff
MaterialsHyper-strong and stiff fiber promises “science fiction” like applications.

“Diamond nanothreads” promise extraordinary properties, including strength and stiffness greater than that of today’s strongest nanotubes and polymers. (Image source: Vincent Crespi lab, Penn State University)
The team—led by John Badding, a chemistry professor at Penn State University and his student Thomas Fitzgibbons—used a specialized large volume high-pressure device to compress benzene up to 200,000 atmospheres. At these pressures, the researchers say, benzene spontaneously polymerizes into a long, thin strands of carbon atoms arranged just like the fundamental unit of diamond’s structure.
The team’s discovery comes after nearly a century of failed attempts by other labs to compress separate carbon-containing molecules, such as liquid benzene, into an ordered, diamond-like nanomaterial.
“We used the large high-pressure Paris-Edinburgh device at Oak Ridge National Laboratory to compress a 6-millimeter-wide amount of benzene—a gigantic amount compared with previous experiments,” said Guthrie. “We discovered that slowly releasing the pressure after sufficient compression at normal room temperature gave the carbon atoms the time they needed to react with each other and to link up in a highly ordered chain of single-file carbon tetrahedrons, forming these diamond-core nanothreads.”
The initial threads created are only a few atoms across or more than 20,000 times smaller than average human hair. According to the team, the next step will be to make the nanothreads at a larger and more practical scale.
http://science.psu.edu
http://carnegiescience.edu